The team led by professor Wang Jian with the laboratory discovers microbiome and metabolome dysbiosis of the gut-lung axis in PH
2022-09-192328Recently, the team led by professor Wang Jian with the laboratory has published an article entitled “Microbiome and metabolome dysbiosis of the gut-lung axis in pulmonary hypertension” in the international journal Microbiological Research (IF=5.070). The study aims to identify the gut microbiota characteristics in the rat model of pulmonary hypertension (PH) established by left pulmonary artery ligation (LPAL), and the interaction between gut microbiome dysbiosis and cecal stool and lung tissue metabolomics, so as to emphasize the previously unknown role of gut microbiome and lung-gut axis in the PH induced by long-term high shear force, and provide new ideas for the treatment, diagnosis or management of PH.
Research background and significance
The intestinal flora alteration can significantly affect the pathophysiological state of the host through multiple mechanisms, including immunological and metabolic changes. Changes in microbial composition and activity in the respiratory tract and gut are known as symbiotic disorders, which are considered to be associated with altered immune responses and the development of lung disease. There have been studies indicating that gut dysbiosis is associated with the disease development of pulmonary hypertension (PH), but most of the current studies are focused on the physiology and pathology of the cardiovascular and pulmonary blood vessels. Therefore, PH should also be considered as a systemic syndrome mediated through multiple-organ interactions, including the potential “gut-lung axis”.
The rat model of left pulmonary artery ligation-induced pulmonary hypertension (LPAL-PH) simulates the pathogenesis of chronic thromboembolic pulmonary hypertension (CTEPH) by increasing the blood flow shear stress and stretching of the right pulmonary artery, providing evidence for the pathogenic link between the microbiota and the lung through the potential “gut-lung axis”, and thus providing a new perspective for the research on the mechanism of LPAL-PH and translational medicine.
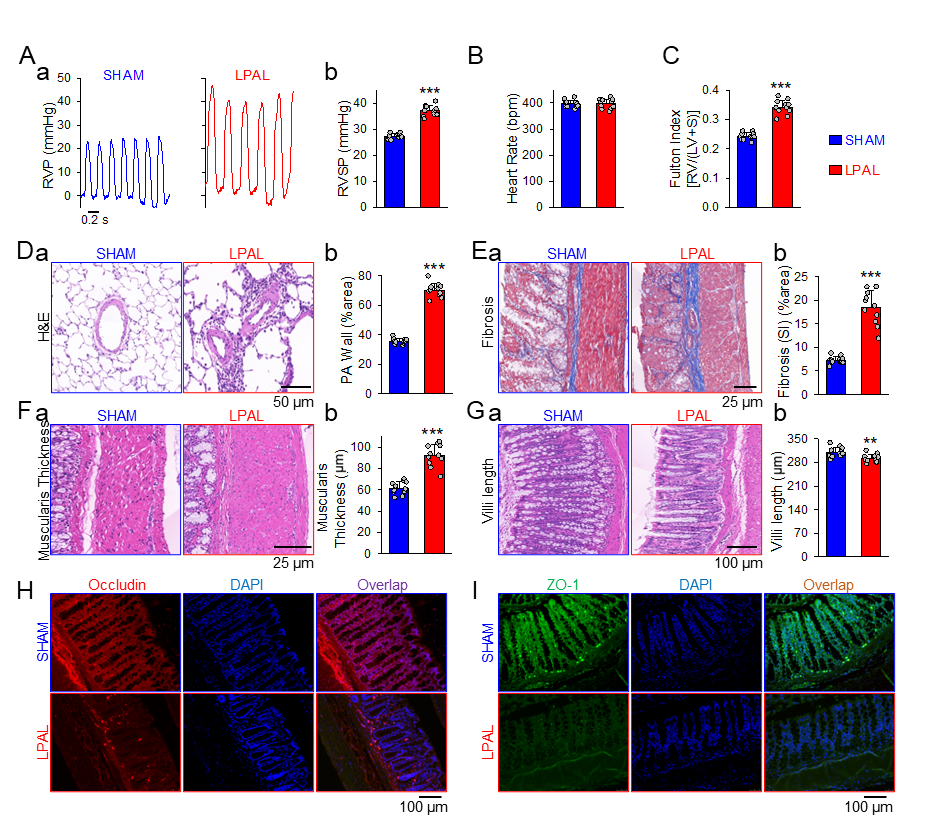
Hemodynamic and lung-gut morphology changes in rat model of left pulmonary artery ligation-induced pulmonary hypertension (LPAL-PH) and sham-operated group
This study is the first attempt to report that the rat model of high shear stress-induced PH is associated with typical pathological changes of gut, such as increased fibrosis, intestinal wall musculature, villus dysplasia, and increased intestinal permeability. The abnormal intestinal pathology is associated with significant changes in the gut microbiome and metabolome, as well as in the lung metabolome. Compared with the sham group, the changes in the intestinal flora of LPAL-PH rats are mainly manifested as increase of Bacillus (Sporobacter), and decrease of eubacterium (Eubacterium), delta protein bacteria (Deltaproteobacteria) and thivibrio (Desulfovibrio). In the gut metabolism group of LPAL-PH and Sham rat groups, the abundance of protective metabolites (such as butyrate and propionate) and pathogenic metabolites (such as proinflammatory mediators) was uneven. Besides, changes in the gut microbiome were closely related to changes of gut and lung metabolomes among LPAL-PH rats. These findings have enriched our understanding of the unique characteristics of the gut microbiome and the close association of the “gut-lung axis” in LPAL-PH induced by long-term high shear stress, providing new ideas for therapy, diagnosis or management of CIEPH.
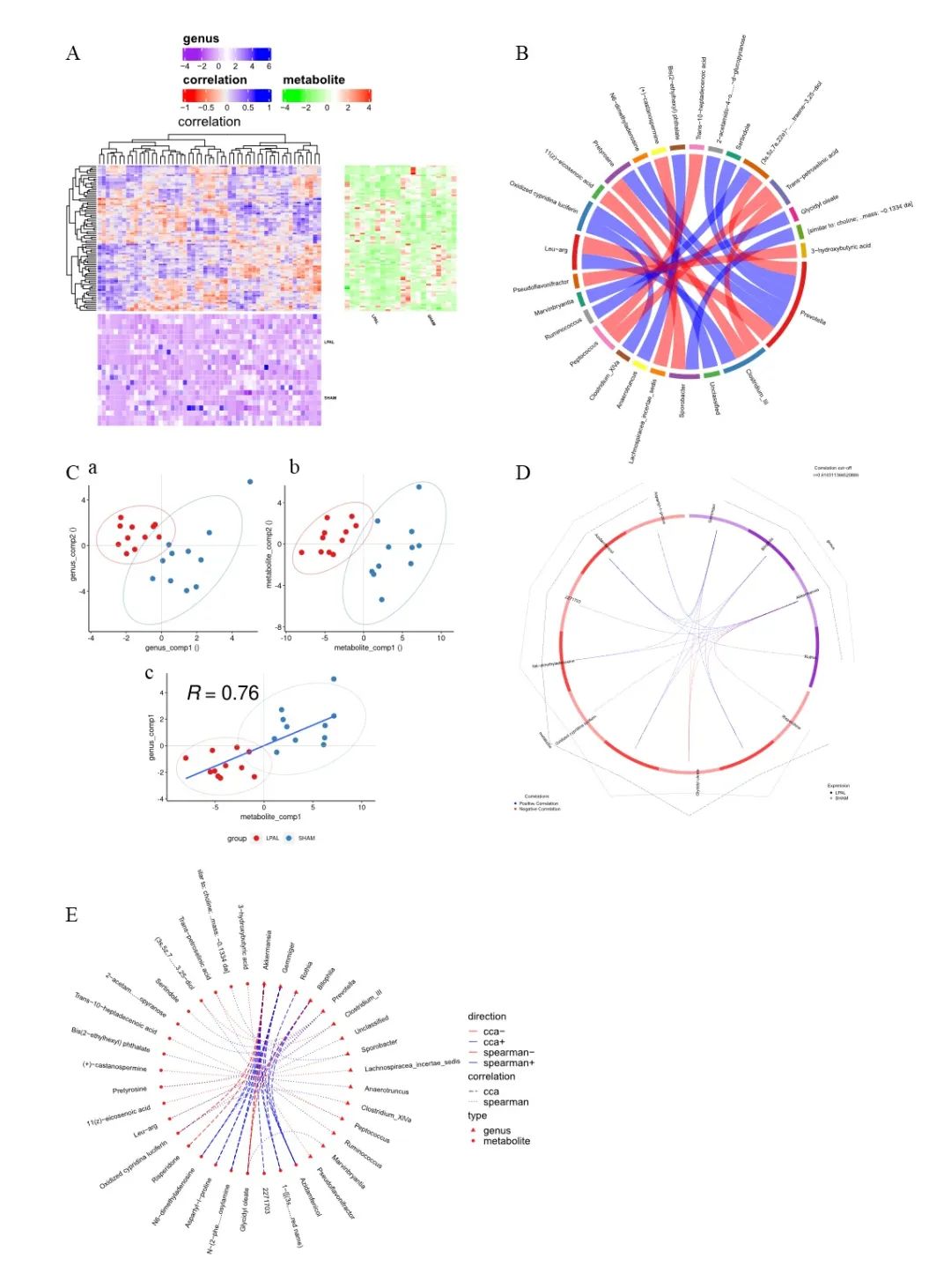
Correlation analysis of gut microbiome and lung metabolomics
















